Search results for #IgAN
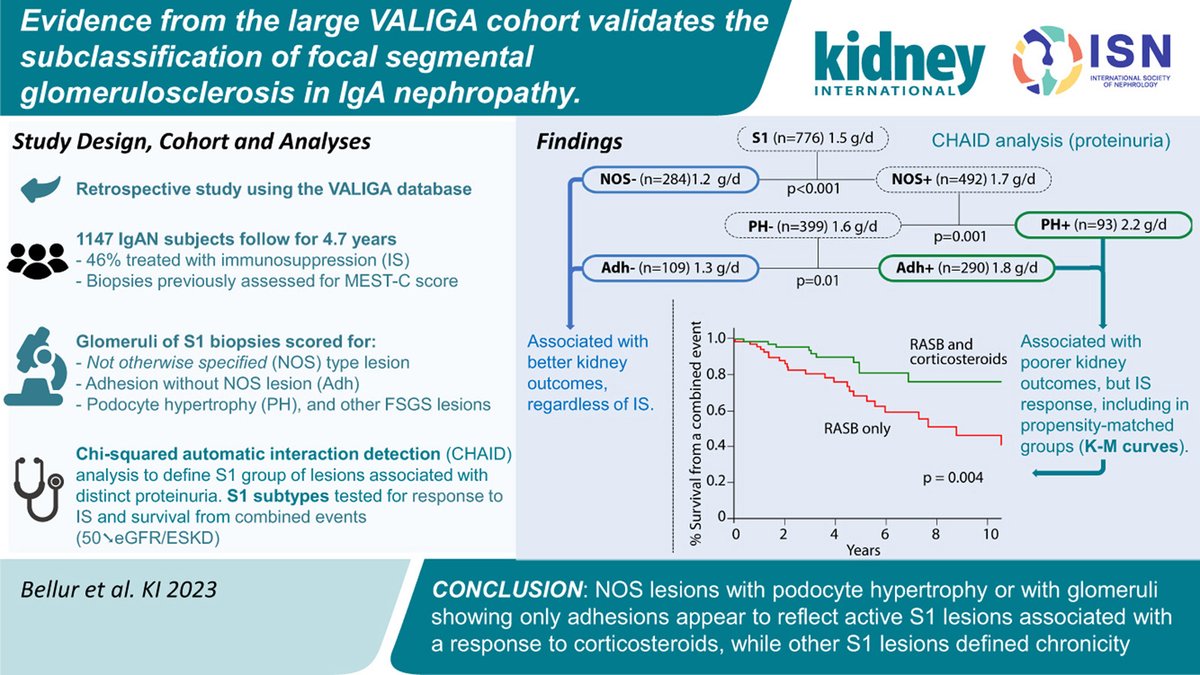
Evidence from the large VALIGA cohort validates the subclassification of focal segmental glomerulosclerosis in #IgA #nephropathy doi.org/10.1016/j.kint… @queensu @UMontreal Oxford University Hospitals NHS Foundation Trust #IgAN #FSGS
A phase 2b, randomized, double-blind, placebo-controlled, clinical trial of #atacicept for treatment of #IgA #nephropathy doi.org/10.1016/j.kint… @StanfordDeptMed @UBCDoM @georgeinstitute @UniklinikAachen @Sydney_Uni @uniofleicester #OpenAccess #IgAN #glomerulardisease
Save the date-spread the word. May 10th, 1800 - Interesting #debates lined up. #IgAN #ANCAVASCULITIS #C3GN #glomerulardisease @isn_india @ISGDtweets
Save the date-spread the word. May 10th, 1800 - Interesting #debates lined up. #IgAN #ANCAVASCULITIS #C3GN #glomerulardisease @isn_india @ISGDtweets
Findings from a recent study highlight the potential value of the light microscopy pattern of glomerular injury and the intensity of mesangial C3 deposition for stratifying #renal outcomes in #IgAN. hcplive.com/view/study-sug…
Learn about the four-hit hypothesis and the role APRIL in IgA Nephropathy in our upcoming webinar. Register today! go.nephu.org/MK4m #IgAN #RareDisease #KidneyHealth #NephU #FourHitHypothesis
Our partner, Everest Medicines, has announced that the Hong Kong Department of Health has approved our treatment for adults with primary IgA nephropathy, expanding access to treatment for #IgAN across Asian territories. Read more: bit.ly/4b02TZQ
In “Recognizing & Addressing Symptom Burden For Patients With IgAN,” a recent NephU article, experts recognize just how influential IgAN is on a patient’s quality of life. Learn more by reading on NephU today! go.nephu.org/MLJX #RareDisease #IgAN #SymptomBurden
Everest Medicines Announces Hong Kong Department of Health Approval of Nefecon for the Treatment for Primary IgA Nephropathy in Adult Patients #ImmunoglobulinANephropathy, #IgAN, #IgANephropathy crweworld.com/article/news-p…
The unmet need of patients with IgA nephropathy has been long underscored by a lack of disease-specific treatment options. With our FDA-approved treatment, patients are now exposed to new possibilities. Watch Chief Medical Officer, Richard Philipson, discuss the #IgAN landscape.
Tune into this webinar to receive a detailed overview of the Four-Hit Hypothesis as well as explain the role of APRIL in disease pathogenesis. Register today! go.nephu.org/MGhf #igan #nephrology #kidneydisease #fourhithypothesis
Want to catch up on the latest in IgAN care? Check out this upcoming LIVE event to hear experts discuss innovations in treatment. Don’t miss out on this comprehensive look at the IgAN therapeutic landscape: ow.ly/a9Xb50Rr18q #MedicalEducation #CME #IgAN #Nephrology
Thank you @ISGDtweets & @isn_india for the invitation to kick off this exciting event & speak about recent clinical trials and new drugs to treat #IgAN Looking forward to a lively discussion!
Thank you @ISGDtweets & @isn_india for the invitation to kick off this exciting event & speak about recent clinical trials and new drugs to treat #IgAN Looking forward to a lively discussion!
Hot Topics in Glomerulnephritis. An online symposium organized by the @isn_india and @ISGDtweets. Session 1 on #IgAN with @SWadhwaniMD @kdjhaveri and @Suceena Register here 👉 us06web.zoom.us/webinar/regist…
Serum Gd-IgA1 was not associated with validated prognostic risk factors but was negatively correlated with kidney function, suggesting further research could detail its utility as a biomarker for risk of #IgAN progression. hcplive.com/view/study-hig…
Did you know the highest prevalence of IgAN is seen in individuals of East Asian descent? Learn more from our infographic, “Kidney Disease In Asian American & Pacific Islanders.” Download now! go.nephu.org/MCqg #IgAN #kidneyhealth #asianamericankidneyhealth #NephU #CKD
Yesterday, @Calliditas1 announced positive results from the NefIgArd open-label extension study of Nefecon in patients with primary #IgAN on optimized RASi therapy. Read more: hcplive.com/view/callidita…
Calliditas Announces Positive NefIgArd Open Label Extension Results in Primary IgA Nephropathy (IgAN) - check the link for more info ow.ly/Jo1450Rnl4i @Calliditas1 # IgANephropathy #IgAN #RareDiseases
Today we announced with CSL Vifor that the European Commission has approved FILSPARI (sparsentan) for the treatment of IgA nephropathy (#IgAN). #InRareForLife Press release: bit.ly/3w9cfUc

Igan Mich @IganMich
2K Followers 552 Following board game designer, entrepreneur, nature lover, from digital back to analog. Invented @Terraternity to create awareness.
Berzan Emrah İgan @Berzannn
55 Followers 4 Following
がんちゃん🐹�.. @igan_yr
478 Followers 150 Following ★ユリ★イェナ★チェウォン★조유리즈 IZ*ONE12人応援しています!幸せでいてほしい…… 坂道など別垢▷@oykr_48 趣味のyoutube▷mineyuu ⚠️プライベートな事も呟きます!
NAGİİİİ @hell_igan
644 Followers 394 Following | Mustafa Kemal ATATÜRK 🌹| ultrAslan AKDENİZ UNI @uAAKDENIZUNI | biblioman | 🌕
IGAN Meitriani @Meitri07
434 Followers 0 Following New Account: @gekmei_ pin: 553CD912 line: gekmeitri ig: meitriii path: Meitriani IGAN SMAN 8 DENPASAR
iGan Partners @iganvc
352 Followers 121 Following Leading Canadian seed stage VC in the Digital Health and MedTech industry. Instragram: @iganpartners
Ayu Partiwi @IganAyu
39 Followers 2 Following Seputar informasi anak? Silahkan ikuti kami di channel youtube kami .....
Igan Veron Entila @IganEntila
184 Followers 89 Following PHILSCANIAN ✈️Aircraft Mechanic ⚒🛩🛠🔩🔧Basketball 🏀/ 💚
𝐏api 𝐈gan, 𝐖.. @rp_thv
2K Followers 2K Following 𝐏𝐀𝐑𝐎𝐃𝐘 ╱ 95‘s pride who have deep voice, bright smile, and zeus visual. Born in Daegu, in charge of being member group called BTS. Spell his name, Kim Taehyung.
ArchitectAyuSulastrin.. @IGANSulastrini
3K Followers 1K Following Ayu Sulastrini for Architecture,Politic,Fashion~Blog:Ayu Sulastrini For Nature,Science,and God~IG: architect_ayu_sulastrini_6810 & IG: as_architects_global
Igan Tinapay @IganTinapay
1K Followers 1K Following I am Igan Tinapay. Explorer in the Latent Space.
Igan Kusniandasari @igankusnianda
690 Followers 208 Following
K(胃がんサバイ�.. @iganSV
159 Followers 25 Following 2018年、38歳で胃がん(印環細胞癌 stage Ⅰa)が見つかり、ロボット支援腹腔鏡下幽門側胃切除術にて胃の2/3を摘出した会社経営者の克服日誌。絶対に病気に勝つ!きっとうまくいく。胃摘出後でも体重減ってないタイプ。最初のツイートから読むと、がん発覚から手術、術後の経過までが分かります。同じ病名ついた人の参考に。
► G♦!!ïG▲N ♫.. @dj_Gi11igan
194 Followers 198 Following Humble youth who enjoys partying ,socializing and offcourse mixing
✨ SHAN ✨ @shan_an_igan
1K Followers 1K Following nintendo & variety streamer 💜 ace 💜 gaymer 💜 DA-2163-7548-7263
PnX Gan3Sh @IGan3sh
546 Followers 204 Following Former Manager of @PNX_eSport (ex iGorillaZ), Pro Club | cb/cdm/cm 🇮🇹 | xbox one 🎮
igan @maeganmnl
474 Followers 541 Following
Dr Igan k peter @igambiirepeter
1K Followers 2K Following MK MOVEMENT co-ordinator Bunyoro Sub-Region,Doctor, political and social Rights Activist, Mk Next President.
igan evanglista @igantarigan
440 Followers 221 Following
Igan @ialarcon21
403 Followers 162 Following †. 21. Ingeniero Agrónomo en potencia, por la Gracia de Dios. Amigo de toda la vida de Super Mario Bros e intimo de Ronald Mcdonals.
umar igan 𝕏 @Umar26338572
134 Followers 618 Following Machine learning, Physics, MSc in Data Science.
erkan igan @ErkanIgan
402 Followers 1K Following
Igan Guerrero @iganguerrero
200 Followers 84 Following Las nubes grises también forman parte del paisaje 🌵🌴🌾
zooligan🇬🇧🇯�.. @IganZool
434 Followers 534 Following Love my Privacy so please do not DM me, Follower of $XRP, $XDC, $XLM and $FLR/ $SGB 👍🏾
리건 @boo1igan
12 Followers 13 Following
MAX IGAN @maxwelligan
3K Followers 2K Following
平八郎@胃がん.. @igan4th_hey86
480 Followers 134 Following 2016.4告知。肝臓に転移したスキルス胃がん(手術不可)、抗がん剤で治療を続けている人。寛解を目指し、したこと、考えたこと、その結果を具体的にブログで公開しています。ツイッターは一緒に暮らす猫、抗がん剤や血液検査、腫瘍マーカーの記録など。
Mich @mich_igan5
250 Followers 284 Following
Oyin_igan🌻 @Moferanola2
337 Followers 301 Following Happy girl😊//BiochemistInEmbryo//lover of God.💛💚//Here for the fun😌
Ganda Raharja @Igan24
66 Followers 0 Following
Igan @prabadikarf
53 Followers 55 Following
Александр К.. @AndrewKoIgan
159 Followers 124 Following
igan erostarbe @ungrateful_79
134 Followers 101 Following these are tough times, and I think most people feel alone in a crowd IP '22
Meiyari A, IGAN @meiyari
282 Followers 220 Following A working at home mom :) Earn money from home with @dBCNetwork http://t.co/ItDKEbyp
igan-iluminacion.com @iganiluminacion
568 Followers 1K Following Tienda de lámparas online e iluminación. Lámparas de techo de marcas como Mantra, Schuller, Faro, Ole by FM y muchas más!!
igan @igan53539570
586 Followers 875 Following
IG/AN @1997IGAN
281 Followers 431 Following![[ ♥ A.A.K.A @goenk_dedik ♥ ]
[ sd 2 saraswati'97 ][ spensa'03 ][ trisma'06'XXX ][ fe.ak.unud'09 ]](https://pbs.twimg.com/profile_images/2959876729/3bdb34762327f3b1029f691a6f7301b3.jpeg)
IGAN Prima Puspa H. @primaimma
214 Followers 513 Following [ ♥ A.A.K.A @goenk_dedik ♥ ] [ sd 2 saraswati'97 ][ spensa'03 ][ trisma'06'XXX ][ fe.ak.unud'09 ]![イグアナ帝国ー!おわったよー!🎶 [Gt.Vo💚@Nana_2167] [Gt.Vo.💙@nemuina_com_] [Ba.💛@oisiimelon_hiwa] [Dr.💜@aa113_0] [Key.❤️@S53Q4PfJeoDDnRN] 運営はぱみーちゃん!!今までありがとうございました🦎](https://pbs.twimg.com/profile_images/1017816230126804993/7qSiXovi.jpg)
イグアナ帝国 @igan_teikoku
139 Followers 188 Following イグアナ帝国ー!おわったよー!🎶 [Gt.Vo💚@Nana_2167] [Gt.Vo.💙@nemuina_com_] [Ba.💛@oisiimelon_hiwa] [Dr.💜@aa113_0] [Key.❤️@S53Q4PfJeoDDnRN] 運営はぱみーちゃん!!今までありがとうございました🦎
philigan @phil_igan
128 Followers 277 Following
irfan igan @irfanigan3
12 Followers 21 Following
igan barker @iganwox
240 Followers 344 Following Pengamat..karena semua orang disini hanya bisa mengetik tanpa menulis, berbicara dan bertindak. I love U Indonesia... GBU
Scare💀igan @KerriganMcknz14
164 Followers 647 Following I make art!! https://t.co/zgTcxlWSE2 Instagram: @thisiskerrigansart she/her ☺️
igan @iganridwan
150 Followers 118 Following 7CCBDD5F || line: iganr, ig: ridwandeska,path: igan ridwan
Lisa Milligan @lisa_mi11igan
72 Followers 143 Following
Bowfish.Igan @BowFishIgan
206 Followers 427 Following
Igan_true blue @igan_respati
264 Followers 550 Following Eat to live, and not live to eat,love traveling, @unas1949 @jakampusUnas TRUE BLUE family and @chelseaindo @CISCdepok @blues tropps cibubur
Thirdeyegang @3rdIGanG7
336 Followers 2K Following
Igan Gandhara @GandharaIg736
21 Followers 15 Following
Igan Suganda @IganSuganda
194 Followers 2K Following
Igan Nothrain @IganNothrain
22 Followers 20 Following
(IGAN)ELITE RESCUE @Philmountaineer
10 Followers 0 Following
CassOwary Igan @CassOwaryE
129 Followers 337 Following Fitness Instructor/C.E.O. & Founder of Flex Dynamics Fitness Solutions & Anamorfit Training Program (IG): @cassowaryhs / @flex_dynamics_fit
たいパパ@胃全�.. @taipapa_igan
204 Followers 223 Following 胃全摘後の食生活や環境の変化について発信します/2022.7.34才/胃がん告知/胃全摘/ステージ1/経過観察/ダンピング症候群/パパ/ママ/子供2才0才/ブログ/デイトラ/web制作/リベ大/つみたてNISA/高配当株/家庭菜園
Antoine M @mich_igan
74 Followers 136 Following